Study of unfolding of the double-knotted protein from Sułkowska group published in eLife
23 02 2026
Category: Main page, Research highlights
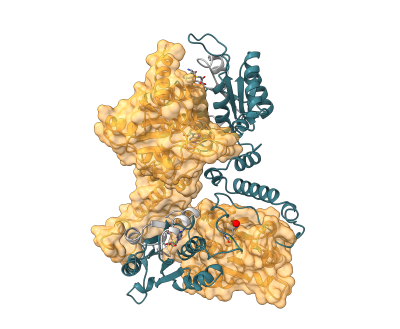
Knotted proteins constitute around 2% of the total entries in the Protein Data Bank (PDB), and so far, only polypeptide chains embedding single-knots have been deposited. In the recent study of the Interdisciplinary Laboratory of Biological Systems Modelling published at eLife journal, CeNT scientists showed the first crystal structure of the double-knotted protein TrmD-Tm1570 from Calditerrivibrio nitroreducens (Cn), PDB ID: 8B1N, belonging to the SPOUT family. It forms a homodimeric complex that binds SAM and Mg2+. TrmD-Tm1570 consists of two domains, TrmD and Tm1570, each of which contains the simplest trefoil knot 31. The detailed structural features of TrmD-Tm1570, as well as those of its individual domains are also presented,
By using biophysical experimental techniques and computer simulations with AI-based methods, Sułkowska group studied their thermal and thermodynamic stability, as well as their mechanical unfolding. Based on molecular dynamics (MD) simulations, with the Structure-Based Cα Model (SBM-Cα) and UNRES (coarse-grained), they showed that native contacts alone are not sufficient to fold double-knotted proteins. However, native contacts are sufficient to fold the single-knotted proteins TrmD and Tm1570 into their native conformations. Using the same model, they identified possible unfolding and untying pathways, in which each domain can self-tie independently. Optical tweezers (OT) experiments showed that this process is also reversible, although the stretched state remains knotted. In addition, they observed higher thermal and mechanical stability in Tm1570 compared with TrmD, which is partly attributable to the position of the knot core. Overall, the results suggest that double-knotted protein from the SPOUT family can only partially self-fold, and that full knotting may require the assistance of a chaperone.
Fernando Bruno da Silva, Szymon Niewieczerza, Iwona Lewandowska, Mateusz Fortunka, Maciej Sikora, Laura-Marie Silbermann, Katarzyna Tych, Joanna I Sulkowska, Multi-barrier unfolding of the double-knotted protein, TrmD–Tm1570, revealed by single-molecule force spectroscopy and molecular dynamics, eLife. https://elifesciences.org/reviewed-preprints/108823