| Project Leader: | Project period: 2017 - 2019 |
| Project funding: POLONEZ, NCN | |
| Project description: Cancer is a complex disease caused by interactions of multiple factors, such as genetic predisposition, environmental and lifestyle influences, infectious agents and ageing. Due to the complexity of this pathology, cancer research includes basic research, strategies for prevention, development of early diagnostic tools, and translational approaches for treatment and cure. In basic cancer research “knowledge” is the key word because if you want to interfere with a biological system you must know how it works. For this reason, the main purpose of this project is to gain information about the mitochondrial form of a DNA repair protein. Radiation therapy and chemotherapy are the mainstream options available for cancer treatment. Many chemotherapeutic drugs act by damaging DNA, leading to an accumulation of lesions that ultimately cause cell death. However, our cells have the ability to repair damages induced by chemotherapeutics drugs therefore vinifying their effects. Base excision repair (BER) is a cellular pathway able to repair the damage generated at nuclear and mitochondrial DNA. An essential protein for the correct functioning of this pathway is APE1. This protein is present both into the nucleus and the mitochondria, represents a key enzyme of the BER pathway and elevated expression levels have been reported in several carcinomas contributing to resistance to chemotherapy. Current approaches to cancer treatment report more effective results when specific DNA repair inhibitors are used in combination with DNA damaging drugs. The foremost rationale of the combined therapy is that the repair of DNA is likely to sensitize cancer cells to chemotherapeutic agents. For this reason APE1’s inhibitors are currently in use in therapy as adjuvant to chemotherapeutic drugs. However, an alternative approach to inhibit APE1’s DNA repair function consists in blocking its mitochondrial translocation to alter the ability of the cell to repair DNA damage induced by chemotherapeutic agents. Unfortunately, information about how APE1 protein reaches the mitochondria are still scanty and contradictory. With this project we intend to better characterize the molecular mechanisms responsible for the translocation into the mitochondrial matrix of the DNA repair protein APE1. This study will significantly contribute to fill the cultural gap about the mitochondrial nature of APE1 protein therefore opening the possibility for further translational approaches for cancer treatment that rely on APE1 protein as a target.
Project disseminations April 18th 2018 Seminar at the Institute of Biochemistry and Biophysics Polish Academy of Sciences, Warsaw. “Unveiling the mitochondrial nature of the DNA repair protein APE1”. Host: Prof. Barbara Tudek. July 8th-13th 2018 Oral presentation at Gordon Research Conference on Mitochondria and Chloroplasts, Lucca (Barga), Italy. “DNA repair protein APE1 degrades damaged abasic mRNA in human mitochondria”. October 3rd 2018 Oral presentation at New Targets in Mitochondria minisymposium, Nencki Institute of Experimental Biology, Warsaw, Poland. “DNA repair protein APE1 degrades damaged abasic mRNA in human mitochondria”. November 28th 2018 Seminar at the University of Freiburg, Freiburg, Germany. “Molecular journey to unveil the mitochondrial nature of a DNA repair protein”. Host: Prof. Bettina Warscheid. June 11th 2019 Popularization activities October 23rd 2017 March 22nd-23rd 2018 and April 24th-25th I organized at the International Preschool of Warsaw (IPW) a straightforward laboratory experience to show to the kids (age 5-6) that:
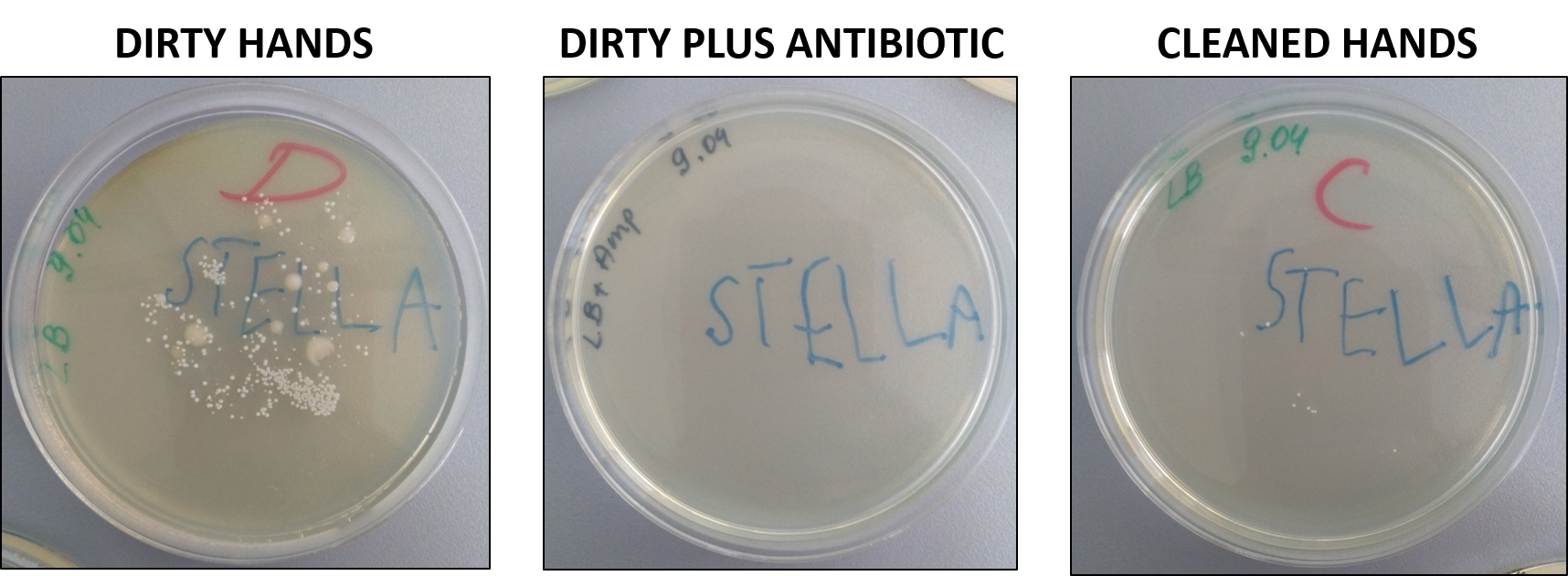
The experience required two consecutive days. During the first day I briefly explained what bacteria are, how they growth, and that some are useful while other are dangerous for us. Then, I explained the experiment. Each kid got three petri dishes: two with solid grow media and one with solid growth media plus an antibiotic. First, each kid touched with his/her hands the petri with only grow media and then the one with also the antibiotic. Then, all kids washed their hands and touched the third petri with grow media. Petri have been incubated for 16h at 37˚C to give time at the bacteria to growth. The day after we observed the results:
This practical experience has been very useful to explain that bacteria are everywhere, and why it is important to wash our hands.
August 30th-31st 2018 I participated at the “Uniwersytetu Młodego Chemika” held at the Centre of New Technologies of the University of Warsaw by holding the seminar “Growing human cells in laboratory to study diseases”. Study visits November 26th-30rd 2018 I spent a week in the laboratory of Prof. Bettina Warscheid at the University of Freiburg. I am collaborating with Prof. Warscheid who is the Head of the laboratory of mass spectrometry in which have been analyzed the samples of APE1’s mitochondrial interacting proteins. During the planned study visit, I will discuss with Prof. Warscheid the progress of the project and organize the rebuttal for the manuscript recently submitted to EMBO Report. During my permanence, I had the opportunity to deepen my knowledge about quantitative mass spectrometry techniques, protein identification, post-translation modifications (PTMs), and protein complex analyses. I also had the opportunity to discuss with Prof. Warscheid abut future scientific collaborations between our laboratories for grant proposal that I plan to apply in 2019. June 11th 2019 I visited the SDS-Optic, a Polish company based in Lublin which develops and creates innovative solutions on the borderline of biochemistry, engineering and fibre optic technologies, that can be used in cancer diagnosis and treatment. Company designed a revolutionary opto-biological methodology that allows for instant cellular processes studies including in-vivo cancer diagnosis with single-cell resolution. During the visit I have been introduced to the different departments (chemistry, biology,…) and also the business office.
Grant application During the period of the fellowship, I applied to the following grants:
Courses attendance During the implementation of the project I attended four courses which were part of the Polonez program. Courses have been organized by the Careers Research and Advisory Centre (CRAC), Cambridge (UK).
Student mentoring During the implementation of the project several of my Italian collaborators joined me at the Centre of New Technologies of the University of Warsaw.
|
|
|
Laboratory of Mitochondrial Biogenesis |
|











 This project has received funding from the European Union’s Horizon 2020 research and innovation programme under the Marie Skłodowska-Curie grant agreement No 665778
This project has received funding from the European Union’s Horizon 2020 research and innovation programme under the Marie Skłodowska-Curie grant agreement No 665778